Medicine Recall: Xarelto 20 mg film-coated tablets (Rivaroxaban)
Strathclyde Pharmaceuticals Ltd is recalling a single batch of Xarelto 20mg film-coated tablets (Rivaroxaban) because of reports of rogue blister strips of 15 mg tablets within two packs of 20 mg tablets. The labelling on one side of the blister strips reflects the correct strength of the tablets as shown in the diagrams.

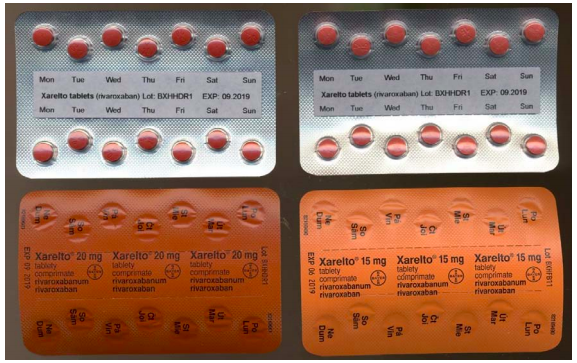
Batch Number:BXHHDR1
Expiry Date: 09/2019
Pack Size: 28 tablets
First Distributed: 18/09/2017
Healthcare Professionals please take the following actions:
- Quarantine any remaining stock from batch BXHHDR1 and return it to the original supplier.
- Identify patients or carers that have received Strathclyde Pharmaceuticals Ltd Xarelto 20 mg tablets (including in compliance aids) since the 18 September 2017 inclusive.
- Contact these patients or carers to notify them of this issue and to ask them whether the tablets have batch BXHHDR1 printed on any of the packaging.
- Ask patients or carers who have received batch BXHHDR1 to check if any of the blisters or tablets are labelled as 15 mg.
- If the patient or carer finds any of the blisters or tablets labelled as 15 mg, they should be asked to return the tablets to the pharmacy where they were purchased or dispensed. If the tablets and blisters inside the carton are the correct strength of 20 mg, patients or carers do not need to return the tablets to the pharmacy.
- If the patient or carer does have tablets of the incorrect strength, make sure that you provide new supplies of this medicine from other batches. It is important for patients not to interrupt their treatment unless instructed by healthcare professionals.
- If patients have been dispensed Strathclyde Pharmaceuticals Ltd Xarelto 20 mg tablets in compliance aids since 18 September 2017 inclusive, patients or carers should be asked to return the compliance aids to the pharmacy in order to be issued with replacements.
More Information on; Medicines and Healthcare products Regulatory Agency
Click to rate this post!
[Total: 0 Average: 0]





1 Comment
BestOllie
August 16, 2019 at 23:08I see you don’t monetize pharmacistdiary.com, don’t waste your
traffic, you can earn additional cash every month with new monetization method.
This is the best adsense alternative for any type
of website (they approve all websites), for more info simply
search in gooogle: murgrabia’s tools